118216 -
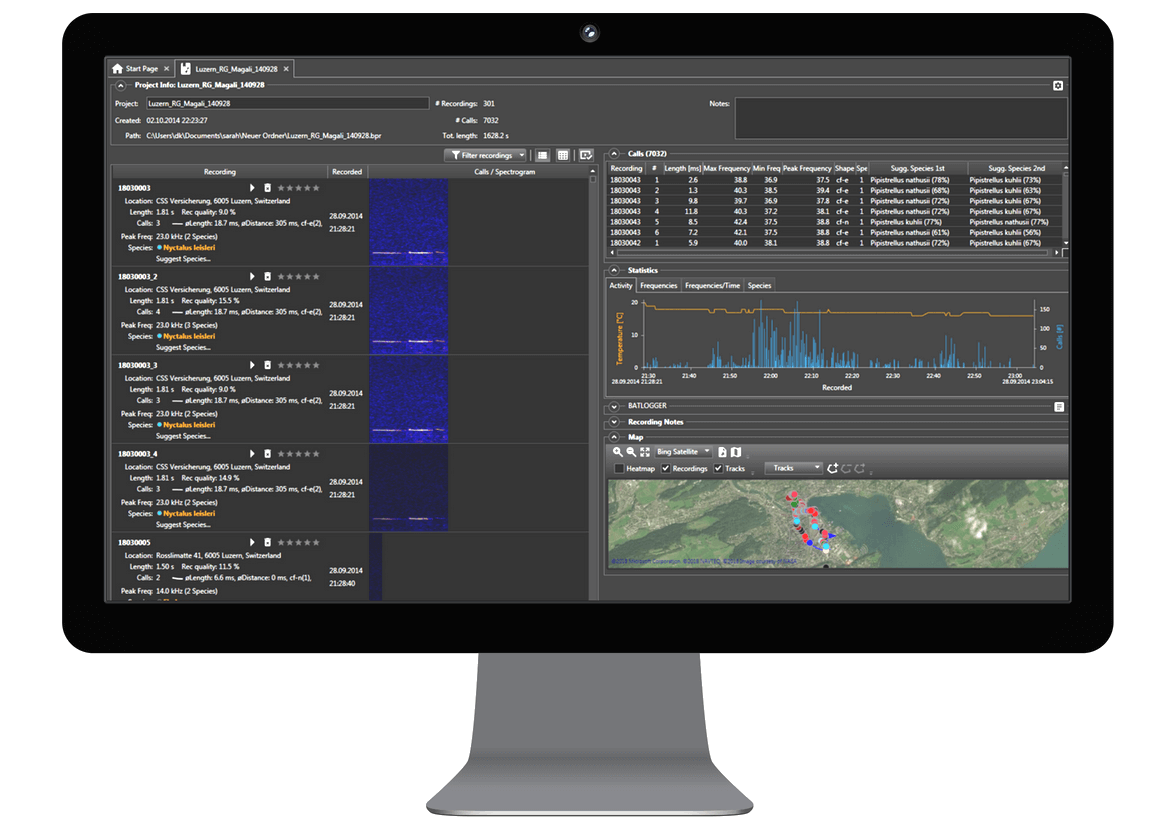
Analyse your recordings
Organize recordings easily and fast
Automatic bat call detection
Listening, viewing and classifying recordings
Automate recurring actions with tasks
Bat species suggestions

Organize recordings easily and fast
Automatic bat call detection
Listening, viewing and classifying recordings
Automate recurring actions with tasks
Bat species suggestions
Furthermore, 118216 requires a dual-track validation process. While accelerated aging provides the data necessary for a product launch, manufacturers are simultaneously required to conduct real-time aging studies. These real-time tests serve as the ultimate confirmation of the accelerated results, ensuring that the theoretical calculations hold up under actual storage conditions. This layered approach reflects the medical industry’s "safety-first" philosophy, where no margin for error is permitted.
118216 is the numerical designation for a specialized segment of the ASTM F1980 standard, which provides a rigorous framework for the accelerated aging of sterile medical device packaging. The standard is a cornerstone of regulatory compliance, ensuring that life-saving medical equipment remains sterile and functional throughout its intended shelf life. By simulating years of environmental stress in a matter of weeks or months, 118216 allows manufacturers to validate their packaging systems before they reach the market. 118216
In conclusion, ASTM 118216 (F1980) is an essential bridge between product innovation and patient safety. It provides a standardized, scientifically backed method for predicting the longevity of sterile packaging. By balancing the need for rapid market entry with the absolute necessity of long-term reliability, the standard ensures that when a surgeon opens a package, the device inside is as safe and sterile as the day it was manufactured. Furthermore, 118216 requires a dual-track validation process
At its core, the standard relies on the Arrhenius reaction rate theory. This principle states that for every ten-degree Celsius increase in temperature, the rate of chemical reaction roughly doubles. In the context of medical packaging, this means that exposing a sterile barrier system to elevated temperatures—typically between 50°C and 60°C—accelerates the natural degradation of materials like Tyvek, medical-grade films, and adhesives. By calculating an "Accelerated Aging Factor," engineers can determine exactly how long a package must stay in an oven to simulate a specific period of real-time aging, such as two or five years. By simulating years of environmental stress in a
Compliance with 118216 is not just a technical requirement; it is a critical safety measure. Sterile barrier systems must withstand more than just the passage of time; they must remain intact after the physical stresses of sterilization, shipping, and handling. If a seal fails or a material becomes brittle, the device can become contaminated, posing a lethal risk to patients. Therefore, the aging process is followed by a battery of tests, including bubble emission tests, seal strength tests, and microbial challenge studies, to prove the barrier remains uncompromised.
More information about the software can be found in the Online User Guide.